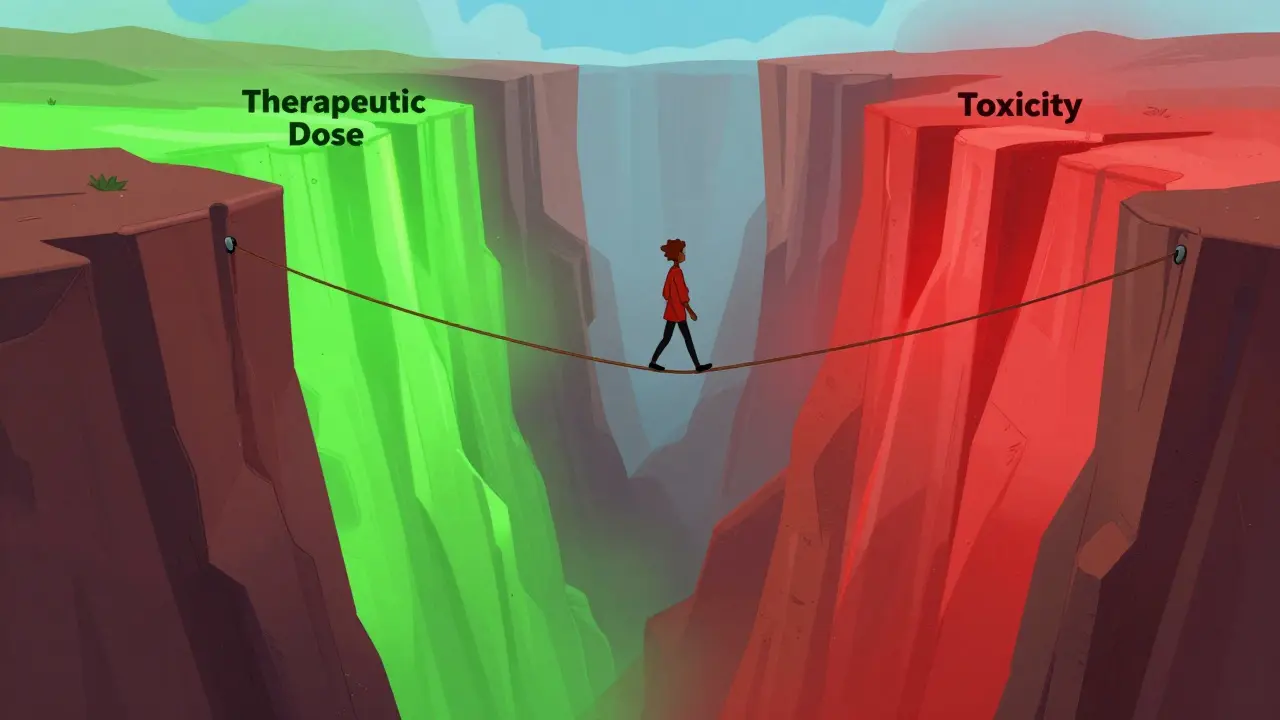
Imagine taking a medication where a tiny change in the dose-something as small as a few milligrams-could be the difference between your treatment working and a trip to the emergency room. This isn't a rare medical anomaly; it's the daily reality for people taking NTI drugs. When you're dealing with a narrow therapeutic window, the margin for error is slim. While switching to a cheaper generic version of a drug usually doesn't cause a stir, doing so with an NTI medication can feel like a gamble for some patients and clinicians.
What exactly are NTI drugs?
A Narrow Therapeutic Index (NTI) drug is a medication where the difference between a dose that works and a dose that becomes toxic is very small . In simpler terms, the "safe zone" for these drugs is tight. If the concentration in your blood drops slightly, the drug stops working; if it rises slightly, you risk severe side effects.
Because of this sensitivity, the FDA (U.S. Food and Drug Administration) holds these drugs to a much higher standard than your average generic. For a typical generic, the active ingredient must fall within a 90-110% range. For NTI drugs, the FDA tightens that window to 95-105%. They also apply stricter bioequivalence rules to ensure that the generic version behaves almost exactly like the brand-name original in the human body.
Common NTI medications you should know
You might be taking an NTI drug without even realizing it. These medications span several different health categories, from heart health to mental stability. Some of the most well-known examples include:
- Heart and Blood: Digoxin (used for heart failure) and Warfarin (a blood thinner).
- Thyroid Health: Levothyroxine (used for hypothyroidism).
- Neurology and Psychiatry: Lithium Carbonate (for bipolar disorder), Phenytoin, and Carbamazepine (for seizures).
- Transplant Support: Tacrolimus and Cyclosporine (immunosuppressants).
Each of these has a different "therapeutic index"-the ratio of a toxic dose to a therapeutic dose. For instance, Digoxin and Tacrolimus both have a ratio of around 2, meaning the toxic dose is only twice the effective dose. That's a very small gap.
The generic switch: Why it's controversial
Here is where it gets tricky. Many patients are fine switching from a brand-name drug to a generic. The real concern arises when a pharmacy switches you from one generic manufacturer to another. This is often done to save costs or because of supply chain issues.
The American Pharmacists Association found that while 87% of pharmacists believe generic NTI drugs are as effective as brand names, 63% have actually heard complaints from patients or doctors about issues after switching between different generic manufacturers. Why does this happen if the FDA says they are bioequivalent? Because "average" equivalence doesn't always account for "individual" variability. Your body might process a version from Manufacturer A slightly differently than a version from Manufacturer B.
| Feature | Conventional Generics | NTI Generics |
|---|---|---|
| Quality Assay Limits | 90% - 110% | 95% - 105% |
| Bioequivalence Range | Standard 80% - 125% | Tighter/Product-Specific |
| Monitoring Requirement | Standard follow-up | Frequent blood tests/TDM |
| Switching Risk | Low | Moderate to High (Individual) |
Real-world examples of switching risks
Let's look at a few specific cases. For people taking Warfarin, the goal is to keep the International Normalized Ratio (INR) between 2 and 3. Research has shown that switching between generic manufacturers can increase the variability of these INR levels. While this hasn't always led to more bleeding events in large studies, it makes the doctor's job of stabilizing the patient much harder.
The situation is even more intense with immunosuppressants. In a study of kidney transplant patients, switching between different formulations of Cyclosporine (specifically Gengraf vs. Neoral) was linked to a 15.3% higher rate of acute organ rejection. When the stakes are an organ transplant, a 15% difference is massive.
Then there's Tacrolimus. This drug has a high coefficient of variation (about 21.9%), meaning the amount of drug in the blood can swing wildly. Different manufacturers have different levels of active ingredients-one might be at 93% while another is at 110%. For a healthy person, this might not matter. For a transplant patient, it could trigger a medical crisis.
How to manage NTI drug switches safely
If you or a loved one are on an NTI medication, you don't have to live in fear, but you should be proactive. The most important tool here is Therapeutic Drug Monitoring (TDM), which involves taking regular blood tests to ensure the drug concentration stays in the safe zone.
Here is a practical approach to handling these medications:
- Ask about the manufacturer: When you pick up your prescription, ask the pharmacist if it is the same manufacturer as your last fill. If it has changed, let them know.
- Document the change: If you feel a difference in how you feel (e.g., a breakthrough seizure or a change in heart rate) after a pharmacy switch, write down the date and the specific generic brand.
- Request "Dispense as Written": If you've had a bad reaction to a switch, your doctor can write "DAW" on the prescription. This tells the pharmacy not to substitute the drug with a different generic.
- Schedule a check-up: If you switch manufacturers, ask your doctor if you need an extra blood test a week or two later to make sure your levels are still stable.
The debate: FDA vs. Clinical Practice
There is a bit of a tug-of-war happening. The FDA maintains that generic NTI drugs are therapeutically equivalent and should be interchangeable. They point to data showing that for drugs like Levothyroxine, nearly 99% of patients maintain stable TSH levels after switching.
On the other side, organizations like the American Academy of Neurology suggest against automatic substitution for certain antiepileptic drugs. They argue that while the "average" patient is fine, the "individual" patient might suffer a breakthrough seizure. This highlights the difference between population-level data (which looks at thousands of people) and clinical experience (which looks at the person sitting in the office).
Are all generic drugs risky to switch?
No, not at all. Most medications have a wide therapeutic index, meaning the gap between an effective dose and a toxic dose is huge. For those, switching generics is generally seamless and safe. The risks discussed here apply specifically to NTI drugs.
Why does my pharmacy change my generic manufacturer?
Pharmacies often switch manufacturers based on cost, availability, or contracts with wholesalers. Since the FDA approves multiple generics as "equivalent," pharmacists generally view them as interchangeable unless the doctor specifies otherwise.
How can I tell if a switch is affecting me?
Watch for a return of your symptoms (lack of efficacy) or new, unexpected side effects (toxicity). For example, if you're on an anti-seizure med and experience a breakthrough seizure, or if you're on a blood thinner and notice unusual bruising, contact your doctor immediately.
Is brand-name always better for NTI drugs?
Not necessarily. Brand-name manufacturers also change their formulations occasionally. The goal isn't necessarily to use the "brand," but to use a consistent version of the drug that your body responds to well.
What is the best way to prevent unwanted switches?
The most effective way is to have your doctor write "Dispense as Written" (DAW) on your prescription. You can also try to use the same pharmacy consistently and ask them to notify you if the manufacturer changes.